 
If it is not available for a specific protein, western blot cannot be used to detect that protein. Primary antibody availability is crucial.Only an approximate estimation and not a precise measurement of molecular weight of the protein is possible Western blot is semi-quantitative at best.Well trained technicians are a must for this technique.This can cause erroneous bands or no bands at all. Insufficient transfer time can result in the larger proteins not transferring properly.The secondary anti-body can sometimes react with a non-intended protein and this can cause labeling of an incorrect protein.A minute imbalance at any level of the procedure can skew the results of the entire process. Western blot is a very delicate and time-consuming process.In the analysis of biomarkers such as hormones, growth factors, and cytokinesĪ few limitations of the Western blot technique are listed below:.In clinical diagnosis – in HIV testing to detect anti-HIV antibody in the serum sample or as confirmatory tests for diseases such as epidermolysis bullosa acquisita or paraneoplastic pemphigus.In the detection of circulating antibodies specific to a single protein or several proteins.Some of the important applications of Western blot are listed below: A labeled secondary antibody which binds to the primary antibody is then added, allowing detection of the specific protein. An egg-derived SARS-CoV-2 vaccine and treatment for COVID-19 diseaseĪ primary or monoclonal antibody is added to the blot, which binds to the target protein. 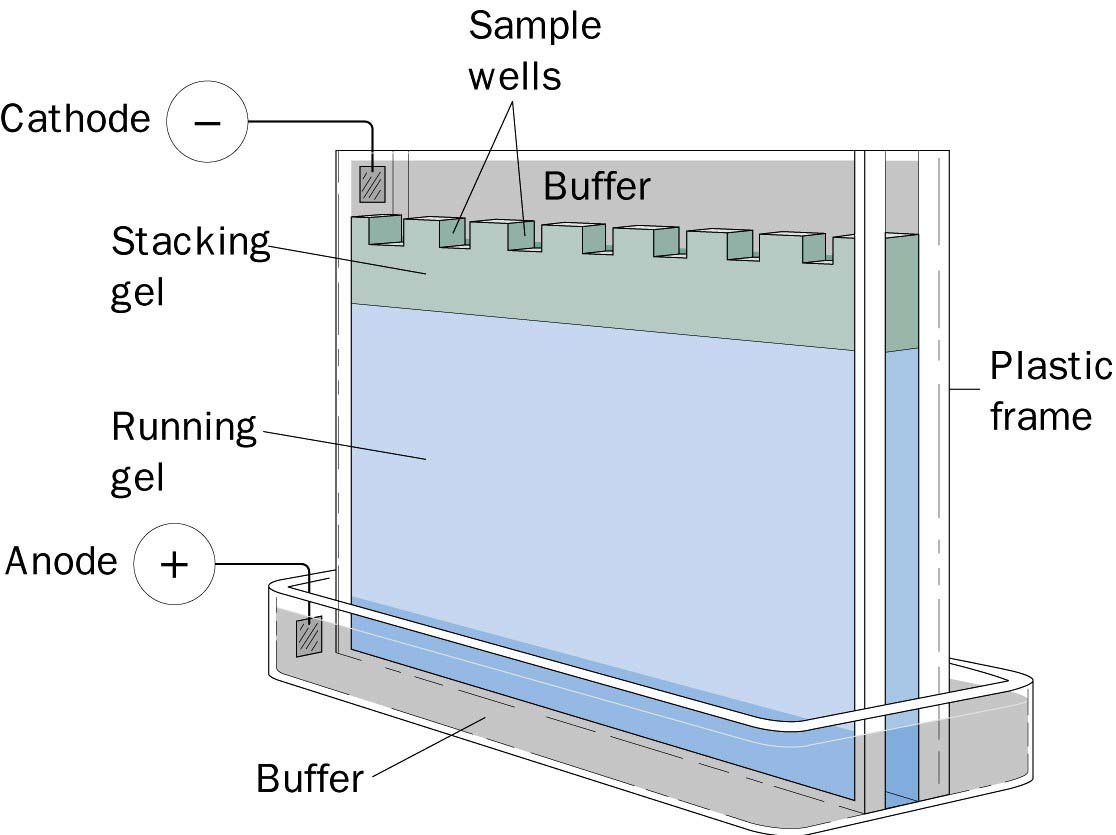
SARS-CoV-2 VOCs dependent on IFITM2 for efficient infection establishment.Scientists find critical role for Hippo signaling pathway in COVID-19 pathogenesis.The pattern of the protein molecules in the gel remains the same in the blotting paper. The separated proteins is transferred to a sheet of blotting paper made of nitrocellulose. Smaller and highly charged molecules move quickly in the gel and go farther than the bulkier molecules. Protein molecules are separated on the basis of their size and electrical charge. The denatured protein sample is loaded onto an electrophoretic gel and an electrical charge is applied. Detergents also offer a negative charge to the protein molecules, which boosts electrophoretic mobility. 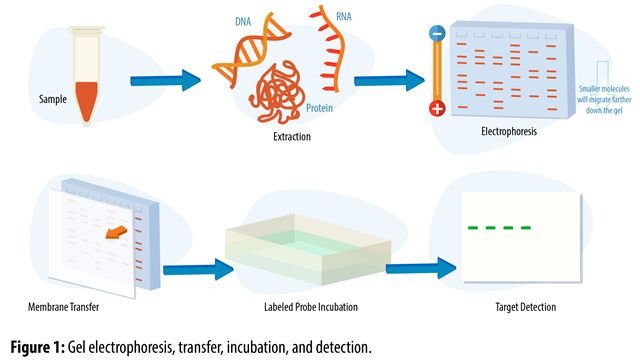
Detergents such as sodium dodecyl sulfate are commonly used to denature proteins. The technique involves 4 key steps, which are discussed below:ĭenaturing involves unfolding of the protein’s tertiary structure to a linear structure. Large protein molecules need to be denatured before electrophoresis to facilitate their movement in the gel. This binding is detected using a chemical or radioactive tag. The separated proteins are then transferred to a solid support, which is then exposed to antibodies that can bind to the target protein. In Western blot, gel electrophoresis is used to separate proteins in a sample based on their molecular weight. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
